This promotional website has been initiated and fully funded by Nutricia. Promotional information regarding Nutricia products will be available on this website.
The Nutricia Allergy range
Nutricia has dedicated over 35 years to advancing the management of CMA to provide a full range of hypoallergenic formulas for your patients with CMA, from birth to 10 years of age.
Delivered to your practice, or
directly to your patient’s home




Helping advance the dietary management of cow's milk allergy in formula-fed infants, SYNEO is the only hypoallergenic formula range to contain synbiotics – a unique blend of prebiotics and probiotics.1*
The SYNEO range includes both Aptamil Pepti Syneo, an Extensively Hydrolysed Formula (EHF), and Neocate Syneo, an Amino Acid-based Formula (AAF).
Between 10%–40% of infants may continue to react to an EHF due to residual cow’s milk peptides in these formulas interacting with the immune system.2,3 An AAF, therefore, may be required.
With Aptamil Pepti SYNEO and Neocate SYNEO, the benefits stack up
- Contains more of the gut microbiota modulating factors found in breastmilk than other UK hypoallergenic formulas1†
- Rebalances the gut microbiota4-9 to support immune development and long-term health10-15
- Shown to support short and long-term clinical benefits8‡-10,16#,17
Our synbiotic blend.* Uniquely combined,1 clinically proven.8-10,16,17

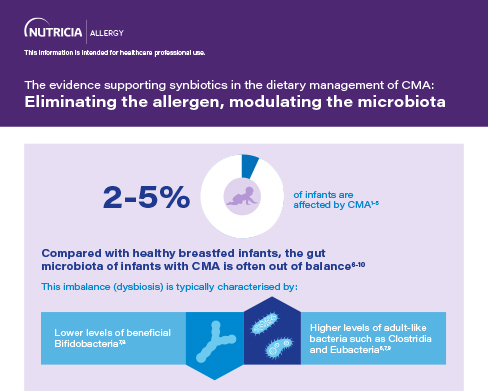
A balanced gut microbiota is important for the development of the immune system.11,12,15 Compared with healthy breastfed infants, the gut microbiota of infants with cow's milk allergy (CMA) is often out of balance.4,6,18-20
Breastmilk is the best source of nutrition for infants with CMA.21,22 Prebiotic oligosaccharides and beneficial probiotic bacteria naturally present in breastmilk support the development of a balanced gut microbiota.23,24
In SYNEO hypoallergenic formulas, synbiotics (prebiotics and probiotics) work synergistically to bring the gut microbiota closer to that of a healthy breastfed infant.4,6-8,25,26
Supported by over 10 years of clinical research, including allergic patients4-10,13,16,17
Research has shown the benefits of supplementing hypoallergenic formulas with synbiotics for the dietary management of cow's milk allergy.4-10,13,16,17
'Compared to non-synbiotic EHFs, Aptamil Pepti Syneo supported reductions in:
- Abdominal discomfort and wind16#
- Constipation and dry stools8‡16#
- Atopic dermatitis severity8‡16#
Compared with a non-synbiotic AAF, Neocate Syneo is associated with:
- 58% fewer skin symptoms17§
- 50% fewer gastrointestinal symptoms17§
- 35% fewer infections17§
- 47% lower rate of antibiotic prescriptions17§
Healthcare professionals and parents report positive experiences with SYNEO27,28
When prescribing Aptamil Pepti Syneo, 100% of GPs said they saw a reduction in GP visits27
"It has greatly improved quality of life for some patients who were really struggling with symptoms"
- Dietitian who had prescribed Aptamil Pepti Syneo27
84% of HCPs reported an observed reduction in the need for medications for GI symptoms28††
"It has made my child a happier and more settled baby"
- Parent of a child prescribed Neocate Syneo28
Using SYNEO in your clinical practice
Click below to request resources, support tools or book a meeting with a Nutricia representative
IMPORTANT NOTICE: Breastfeeding is best. Aptamil Pepti Syneo, Neocate Syneo, Aptamil Pepti 1, Aptamil Pepti 2, Neocate LCP, Neocate Spoon, Neocate Junior are foods for special medical purposes. They should only be used under medical supervision, after full consideration of the feeding options available including breastfeeding. Aptamil Pepti Syneo, Aptamil Pepti 1, Aptamil Pepti 2 are used for the dietary management of cow’s milk allergy. Aptamil Pepti Syneo and Aptamil Pepti 1 are suitable for use as the sole source of nutrition for infants from birth, and/or as part of a balanced diet from 6 months. Aptamil Pepti 2 is suitable for use for infants from 6 months as part of a balanced diet and as a principal source of nourishment with other foods for children. Neocate Syneo, Neocate LCP, Neocate Spoon and Neocate Junior are used for the dietary management of cow’s milk allergy, multiple food protein allergies and other conditions where an amino acid-based formula is recommended. Neocate Syneo and Neocate LCP are suitable for use as the sole source of nutrition for infants under one year of age. Refer to label for details.
* SYNEO synbiotic blend: Bifidobacterium breve M-16V (probiotic) & short and long-chain galacto- and/or fructo-oligosaccharides (prebiotic). The only synbiotic blend within hypoallergenic formulas in the UK.
†Aptamil Pepti Syneo and Neocate Syneo contain more of the gut microbiota modulating factors found in breastmilk (Prebiotic oligosaccharides [short and long-chain galacto- and/or fructo-oligosacharides], probiotic bacteria [Bifidobacterium breve M-16V] and lactose [Aptamil Pepti Syneo only]) than other UK hypoallergenic formulas.
‡12-week RCT8. Infants with atopic dermatitis, Aptamil Pepti Syneo vs non-synbiotic EHF. Subgroup of n=50 infants with IgE-associated AD, difference in SCORAD score Aptamil Pepti Syneo vs non-synbiotic EHF, -4.6, 95% CI, p=0.04.
#Single arm UK non-IgE-mediated CMA study,16 baseline non-synbiotic formula (n=27 out of n=29 well established on a non-synbiotic EHF) vs Aptamil Pepti Syneo, 4-week intervention. Sig reduction in GI symptoms (abdominal pain, wind, & constipation p=<0.05), reduction in severity of rhinitis (p<0.05) and itchy eyes (p<0.05). Significant reduction in PO-SCORAD in subgroup of n=6 infants with more severe AD at baseline, p=0.03. Mean number of overall hospital visits and hospital medication prescriptions significantly reduced (p<0.05) in the 6 months after Aptamil Pepti Syneo initiation compared with 6 months prior. n=13 included in the follow-up analysis.
§Observational study of real-world evidence in The Health Improvement Network (THIN) GP database16, n=148 Neocate Syneo vs Alfamino (Feb 2021).
†† Reported change since starting Neocate Syneo, parent & HCP survey, n=120 HCPs n=50 parents, Jan 2023. HCPs recorded when they always, often or sometimes noticed a reduction.
‡‡Product can be provided to patients upon the request of a healthcare professional. They are intended for the purpose of professional evaluation only.
AAF: amino acid-based formula; AD: atopic dermatitis; CMA: cow’s milk allergy; EHF: extensively hydrolysed formula; GI: gastrointestinal; HCP: healthcare professional; PO-SCORAD: Patient‐Orientated SCORing AD; RCT: randomised controlled trial; THIN: The Health Improvement Network
References
1. Market comparison of UK EHF and AAF data cards, February 2024.
2. Meyer, et al. J Allergy Clin Immunol. 2018;6(2):383-99.
3. Hill, et al. Clin Exp Allergy. 2007;37(6):808-822.
4. Candy, et al. Pediatr Res. 2018;83(3):677–86.
5. Abrahamse-Berkeveld, et al. J Nutr Sci. 2016;5:e42.
6. Burks, et al. Pediatr Allergy Immunol.2015;26(4):316–22.
7. Fox, et al. Clin Transl Allergy. 2019;9:5.
8. van der Aa, et al. Clin Exp Allergy. 2010;40(5):795–804.
9. Sorensen, et al. Nutrients. 2021;13(3):935.
10. van der Aa, et al. Allergy. 2011;66(2):170-7.
11. Martin, et al. Benef Microbes. 2010;1(4):367–82.
12. Wopereis, et al. Pediatr Allergy Immunol. 2014;25(5):428–38.
13. Harvey, et al. Pediatr Res. 2014;75(2):343–51.
14. Walker, et al. Pediatr Res. 2015;77(1):220–8.
15. West, et al. J Allergy Clin Immunol. 2015;135(1):3-13.
16. Hubbard, et al. Immun Inflamm Dis. 2022;10(6):e636.
17. Sorensen, et al. Nutrients. 2021;13(7):2205.
18. Canani, et al. ISME J. 2016;10(3):742-50.
19. Harmsen, et al. JPGN. 2000;30(1):61-7.
20. Thompson-Chagoyan, et al. Pediatr Allergy Immunol. 2010;21:e394-400.
21. WHO. Infant and young child feeding, 2003.
22. Vandenplas, et al. J Asthma Allergy. 2021;14:1243-56.
23. Moossavi, et al. Front Pediatr. 2018;6:197.
24. Boehm, et al. J Nutr. 2007;137(3Suppl.2):847S-9S.
25. Pandey, et al. J Food Sci Technol. 2015;52(12):7577-87.
26. Chua, et al. J Pediatr Gastroenteral Nutr. 2017;65(1):102-106.
27. Data on file. Aptamil Pepti Syneo parent and HCP experience survey, n=20 HCP, n=10 parents. March 2022.
28. Data on file. HCP and parent survey, n=120 HCPs, n=50 parents. Jan 2023.